|
This article may be reprinted free of charge provided 1) that there is clear attribution to the Orthomolecular Medicine News Service, and 2) that both the OMNS free subscription link http://orthomolecular.org/subscribe.html and also the OMNS archive link http://orthomolecular.org/resources/omns/index.shtml are included.
|
|||||||||||||||||||||||
|
FOR IMMEDIATE RELEASE
IMPACT OF INTRAVENOUS VITAMIN C MEGADOSE ON THE QUALITY OF LIFE OF TERMINAL CANCER PATIENTSby Hugo Galindo Salom, Carlos Carrillo and Gladys Galvis(OMNS August 26, 2020) This observational study was conducted to assess the impact of intravenous megadose vitamin C on the quality of life (QoL) of terminal cancer outpatients experiencing side effects of cancer therapies (radiotherapy, chemotherapy, surgery and/or hormonotherapy). MethodsWe asked doctors in two medical centers in Bogota in Colombia to identify cancer patients who are both experiencing side effects from conventional treatments and have been prescribed high dose intravenous vitamin C (HDIVC) as part of their treatment. The HDIVC protocol used with terminal cancer patients in the Bogota medical centers uses 140 grams of HDIVC over 7 days. The medical centre doctors explained the study to identified patients, sought consent, and asked patients to fill out the European Organization for Research and Treatment of Cancer questionnaire EORTC-QLQ C30 version 3 (QLQ-30) before their HDIVC treatment. 22 patients were entered into the study, 19 completed. Our observational study compared the pre and post HDIVC QLQ-30 data for these 19 patients. The collected QLQ-30 raw data was scored according to EORTC guidelines and also arranged into 5 categories; Domestic activity, Everyday (Routine) activity, Emotional activity and General QoL (Patient Score). The summed results of the raw data for each of these categories are presented and a percentage change for QoL results after HDIVC treatment is given. ResultsFor the QLQ-30 scale scores significant improvements in QoL (p < 0.01) were observed for: Global health status/QoL, Physical functioning, Role functioning, Emotional functioning, Social functioning, Fatigue, Pain, Insomnia, and significant improvements in QoL (p < 0.05) were observed for: Nausea and vomiting, Dyspnoea, Appetite loss. For the raw data the mean ± SD percentage change improvements in QoL observed were: Domestic activity (20.99% ±33.53%), Routine activity (30.98% ±22.40%), Emotional activity (33.22% ±21.18%), Sum of Domestic + Routine + Emotional (31.49% ±18.21%), General QoL (100.21% ±140.86%). ConclusionsOverall, the application of high dose intravenous vitamin C had a significant positive effect on patients' quality of life in all categories. No significant side effects to high dose intravenous Vitamin C administration were reported. BackgroundA significant literature supports the relationship between pharmacological consumption of vitamin C and the health of animals and humans (1). It is now well established that megadoses of Vitamin C have functions well beyond its early classic use in treating scurvy. It has well established roles in the formation of collagen (2), cholesterol (3) some hormones (4), and the epigenetic switching of methylated cytosine in the promoter region of genes (5). Ascorbic acid also acts as a strong antioxidant and free radical scavenger decreasing inflammation in numerous disease processes including sepsis (6), burns (7) and cancer (8). It also increases the absorption of inorganic iron through reduction of ferric ion to ferrous (9). A typical HDIVC dose used in treatment and research for over two decades has been 60 grams (10). More recently a wide variety of doses and dose schedules have been used. A recent review (11) details several studies examining multiple doses and dose schedules that show that Vitamin C has significant effects on inflammation in cancer, and that Vitamin C is typically very depleted in cancer patients undergoing standard treatments. The use of HDIVC in conjunction with medical cancer treatments in clinical research and treatment is now becoming widespread. A recent review (12) discusses Vitamin C deficiency in cancer, benefits in QoL and management of side effects with HDIVC in cancer patients, reduction in inflammation in cancer patients, and reviews dose ranges and some of the understood mechanisms for HDIVC in cancer. Since the proposal by the twice Nobel Prize winner Linus Pauling about the efficacy and necessity of administering megadose Vitamin C intravenously (approx. 10 grams per dose) - rather than just orally - to improve the outcome of cancer patients, researchers studying Vitamin C in cancer have continued seeking to find the appropriate doses of vitamin C, when delivered intravenously in this pathology (13). The patient cohort included in the Pauling and Cameron cancer study had not received any significant cytotoxic therapy nor radiotherapy which may have resulted in more significant immune stimulation after megadose intravenous Vitamin C (14). The pharmacokinetics of higher concentrations of Vitamin C achievable in the blood stream through intravenous administration compared with oral administration are well established (15). The Quality of life (QoL) concept first appeared in 1948, when the World Health Organization (WHO) defined "health" as complete physical, mental and social well-being; superseding an older concept of health as the absence of disease or infirmity. The current definition, although criticized because of the difficulty of defining and measuring well-being, remains an ideal. Later, the term quality of life (QoL) evolved from a purely conceptual definition to a series of scales. This new definition measures the general perception of the individual. A commonly used tool to measure QoL in cancer patients is the European Organisation for Research and Treatment of Cancer questionnaire EORTC-QLQ C30 version 3 (QLQ-30) (16). The QLQ-30 is scored into scales that measure functionality, pain and disability and emotional scales that are influenced by personal experiences and the expectations of a person (17). From several previous studies is apparent that high dose vitamin C improves QoL in cancer patients. Cameron and Pauling in 1976 (14) demonstrated that megadose intravenous Vitamin C (typical dose 10 g) along with oral Vitamin C not only improved the quality of life of cancer patients but on average increased the length of survival. Vollbracht et al. in 2011 (18) studied the effect of 7.5 g of intravenous Vitamin C weekly on the QoL of cancer patients receiving standard tumor therapy and aftercare. The intensity score for symptoms was close to twice as high in the placebo group compared to the IVC group. Ou et al in 2017 (19) conducted a pharmacokinetic and QoL study in China on patients receiving electro-hyperthermia along with high dose intravenous Vitamin C for advanced non-small cell lung cancer. They saw significant improvements in EORTC QLQ-30 physical function scores and symptom scores for all high dose ranges of intravenous Vitamin C tested; 1.0 g/kg, 1.2 g/kg and 1.5 g/kg - 60 to 90 grams of Vitamin C for a 60 kg person. Takahashi et al. (20) in 2012 observed the use of a high dose Riordan protocol - dose adjusted after the third day of IVC treatment for each patient to reach a plasma Vitamin C concentration of 350 mg%, typically approx. 140 g IVC per week. The study was conducted in multiple centres in Japan and saw dramatic increases in overall EORTC QLQ-30 QoL scores in a group of 60 patients after 2 and 4 weeks of HDIVC treatment. Yeom et al. in 2007 (21) used 10 g IVC in 2 doses 3 days apart in cancer patients and measured the change in QoL with the QLQ-30 questionnaire. They found a significant improvement in QoL in multiple function and symptom scales. Carr et al. (11) reviewed several trials and case studies reporting positive effects by HDIVC on QOL in cancer patients with or without chemotherapy. They note that the typical limitations in QoL studies of HDIVC in cancer are: the studies do not use a placebo control, the studies do not examine dose ranging effects, and the studies do not measure the duration of effectiveness of a dose. Bazzan et al. in 2018 (22) retrospectively examined the effects of HDIVC on 86 patients of the Thomas Jefferson University Hospital over a 7-year period. They found that overall HDIVC was safe, well tolerated and was effective at improving QoL for these patients. Vitamin C megadose therapy has been safely used for over 50 years in developed countries as a complementary medical treatment for cancer. We prospectively observed the effects of intravenous vitamin C therapy on the quality of life in a group of Colombian patients with advanced cancer, for whom conventional oncological treatments were producing side effects. Our aim was to observe the effects of high dose Vitamin C on our population and see how it compares with other results from around the world. METHODSThis is an observational, analytical, prospective and comparative study (patients fill out a questionnaire both pre and post treatment). The questionnaire is the European organization for research and treatment of cancer quality life EORTC QLQ-C30 version 3 (16). Human Right Statements and informed consentAll procedures followed were in accordance with the ethical standards of the Helsinki Declaration of 1964 and its later amendments. Informed consent was obtained from all patients for being included in the study. COLLECTION OF BASIC INFORMATION (Patient Questionnaire)The most validated method used today worldwide to measure QoL has been developed by the European Organization for Research and Treatment of Cancer and is called EORTC QLQ-C30 version 3 (16). The EORTC is an international organization in which investigators from different countries participate in cancer research; in this case with a special emphasis on QoL of patients who suffer from cancer (17). Our reference population is adult outpatients in the city of Bogota (Colombia) with any malignant cancer diagnosis receiving conventional treatments including chemotherapy, radiotherapy and/or hormone/therapy. Suitability for treatment and consentAll patients receiving HDIVC at the Bogota medical centres are tested that they are medically fit to receive HDIVC prior to treatment. This includes checking for contraindications and completing tests for a) Serum chemistry profile with electrolytes, b) Complete blood count (CBC) with differential and c) Red blood cell G6PD (must be normal). All patients receiving HDIVC have signed consent forms. We asked the medical centre doctors to identify suitable outpatients according to inclusion and exclusion criteria: Inclusion CriteriaAdult (18 years old or older) External ambulatory patient (outpatient) with a 5 year (or less) active malignant cancer diagnosis being treated with chemo, radio, hormone therapy, or surgery. Individual who can read, understand and answer the EORTC QLQ-C30 questionnaire version 3. No evidence of G6PD deficiency or abnormal kidney function. Exclusion CriteriaUnder 18 years old. Be hospitalized (inpatient) Not being treated with chemotherapy, radiotherapy and/or hormone therapy. Not able to read, understand and answer EORTC QLQ-C30 questionnaire version 3. Patients with the following cancers were recruited into our study; Breast cancer, myoepithelial carcinoma, ovarian cancer, kidney carcinoma, non-Hodgkin's lymphoma, pleural mesothelioma, pleomorphic sarcoma, gastric carcinoma, stomach adenocarcinoma, brain macroadenoma, lung cancer, frontal lobe astrocytoma, transverse bowel cancer, ductal adenocarcinoma, bladder carcinoma, womb cancer INTRAVENOUS VITAMIN C USED BY THE BOGOTA CLINICS:Vitamin C as Sodium Ascorbate solution. Each vial contains 100mL, with 11.2 grams of Sodium Ascorbate for injection equivalent to 10 grams ascorbic acid. Manufacturing laboratory: Biological Therapies, Victoria Australia Registered in Colombia INVIMA 2016M-0012358-R1 In the Bogota medical centres cancer patients who receive HDIVC are given a standard protocol. The protocol is based on the work of Drs. Riordan and Hunninghake et al. (23) but is modified for use in Colombia. The protocol we observed was used by the medical centre doctors (H. Prieto, S. Rojas, R. Leudo) trained in injectable nutrients therapies. An online version of the original Riordan protocol is maintained by the Riordan Clinic (24). It states that "Research and experience have shown that a therapeutic goal of reaching a peak-plasma concentration of ~20 mM (350-400 mg/dL) is most efficacious. (No increased toxicity for post-IVC plasma vitamin C levels up to 780 mg/dL has been observed.) The first post-IVC plasma level following the 15-gram IVC has been shown to be clinically instructive: levels below 100 mg/dL correlate with higher levels of existent oxidative stress, presumably from higher tumor burden, chemo/radiation damage, hidden infection, or other oxidative insult, such as smoking." In Colombia post-IVC plasma vitamin C levels measurements to determine the oxidative burden are not available (at May 15th, 2016). Since 2013 medical centre doctors have been utilizing the European patented Vit C test (Free Radical Analytical System 4, Evolvo Italy TM); It is relevant to mention that the Regulatory Body in Colombia, INVIMA, has only approved a concentration of 100mg/mL for IVC. For the above-mentioned reasons the American Riordan Protocol has been adjusted to Colombian conditions (test and concentration). ProductsSodium Ascorbate Solution: vial 11.2 g in 100 ML (equivalent to 10 gr of Ascorbic Acid)
100 ml of Water for Injection. Magnesium Sulfate: 10 ml ampoule of Mg So4 al 20% (2.5 gr x 10 ML)
Administration of IVC (dosages): 14 vials = 140grams in 5 dosesContraindications, precautions and potential side effects are outlined in detail in Riordan´s Protocol (24). After completing an oxidative stress test (Free Radical Analytical System, FRAS 4) and after patients have completed the Pre-HDIVC QLQ-30 questionnaire, the administering physician begins with a series of three consecutive IVC infusions at 15, 25, and 50-gram dosages. Following the first three IVCs, the patient is scheduled to continue with a 25-gram IVC dose twice a week. Day 1:
Day 2:
Day 3:
Day 4: (3 days after day 3)
Day 5: (2 days after Day 4)
Final course of action: Oxidative stress test completed (FRAS4), Post-HDIVC QLQ C30 questionnaire Note= Oral vit C 1 gram every 6 hours during break days after Day 3 is advisable. The post HDIVC questionnaire was answered by 19 outpatients. The outpatients' quality of life (QoL) was measured using the QLQ-C30 questionnaire, both three days before and three days after application of HDIVC. ENTERING THE QLQ-30 DATA INTO DATABASE CATEGORIESThe QLQ C-30 TABLE A questionnaire was converted into specific category groups for entry into a database: Domestic Activity.
DATABASE VARIABLES62 variables are defined as follow: Start: variable 1 and variable 2 describe the patient as pre QLQ-30 or post QLQ-30 General Data: Covers the identification variables of the patients - diagnosis - status - value of laboratory tests used by external doctors (Pharmanex Biophotonic Scanner 3 and Free Radical Analytical System, FRAS 4). Variables 3 to 11. Inclusion Criteria: Covers variables 12 to 18 and describes how the inclusion criteria are fulfilled. Exclusion Criteria: Covers variables 19 to 26 and describes how the exclusion criteria are fulfilled. Date EORT: variable 27 specifies when (the date) EORT was taken pre and post application of the Riordan protocol Domestic Activity: Covers variables 28 to 33 which include the first five questions of the QLQ-C30 that categorized as "Domestic Activity". Variable 33 describes the total score of this category. Every day (Routine) Activity: Covers variables 34 to 48 which include questions 6 to 19 of the QLQ_C30 that are categorized as "Routine Activity". Variable 48 describes the total score to this category. Emotional activity: Covers variables 49 to 59 of the QLQ-C30 that are categorized as "Emotional Activity". Variable 58 describes the total score of this category, variable 59 is the total score of the 3 categories. General evaluation: variables 60 to 62 "General evaluation". Variable 62 describes the total score of this category. All patients fulfilled the inclusion / criteria. Scoring the QLQ-30The European Organization for Research and Treatment of Cancer has published procedures for scoring the QLQ-30 (25). We have presented the Scored data (the QLQ-30 Scales) for pre and post HDIVC and also presented a percent change statistic calculated on the raw data for pre and post HDIVC. QLQ-30 scalesThe procedure is to combine the QLQ-30 data into 15 scales, calculate a raw score for each scale (the average of all elements in the scale), then calculate an adjusted score ranging from 0 - 100 for each scale (the "Score"). In the QLQ-30 scores the following apply:
For the general QoL scale a higher score = a higher QoL
Raw dataA scale with a score ranging from 0 - 100 is not suitable for our percent change descriptive statistic. The adjusted Score for each patient will on several occasions produce a value of zero which produces a "divide by zero" error in percent change calculations. Rather than adjust this further we chose to use the raw data only for our percent change calculations. The raw data will never contain a zero. Data presented in our 5 categories; Domestic activity, Routine activity, Emotional activity, Sum of these three, and the General QoL are all sums of raw data. Questions 1 - 28 in the QLQ-30 all have 4 possible responses:
For questions 1 - 28 a low number in the raw data represents a higher QoL. Questions 29 - 30 in the QLQ-30 all have 7 possible responses:
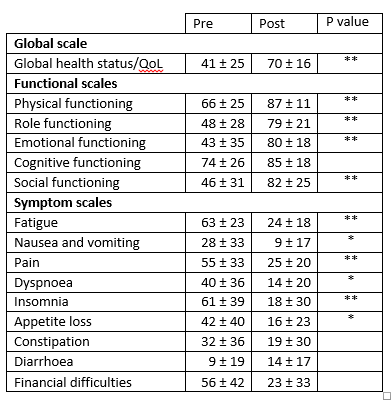
For these two questions a high number in the raw data represents a higher QoL. RESULTSNo significant side effects to HDIVC administration were observed. QLQ-30 ScoresThe QLQ-30 data was sored according to the EORTC manual (25). Wilcoxon signed rank tests were used to determine statistical significance. Table 1: Results. QLQ-30 scores HDIVC 140 g in 1 week EORTC QLQ-30 Score ± SD
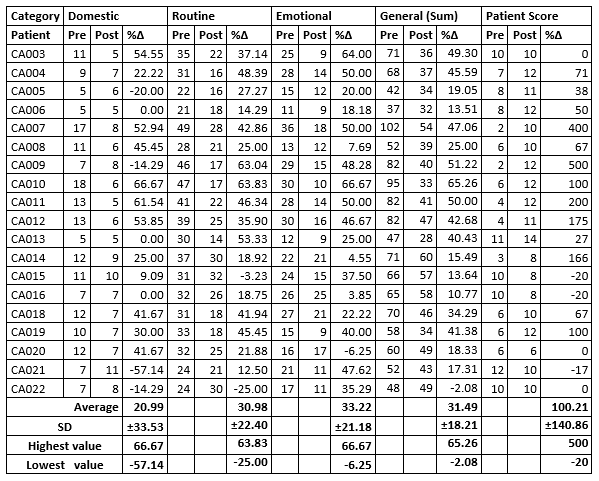
Significance: * P < 0.05 ** p < 0.01 DESCRIPTIVE STATISTICS (Percent Change)Domestic Activity (4 possible responses for each question): Our category Domestic Activity covers the first 5 questions (1 to 5) of the QLQ-C30 questionnaire. Each of these questions has a range from 1 - 4, so the possible sum of the raw data ranges from 5 - 20, where 5 is the highest QoL (excellent QoL), and 20 is the lowest QoL (very poor QoL). In our Domestic Activity category, we observed that 4 out of 19 patients replied that their QoL was impaired (20.99%), 3 patients replied their QoL did not change at all (15.78%), and 12 patients replied their QoL improved (63.15%). The greatest improvement in QoL was 66.67%; and the greatest decrease in QoL was 57.14%; with a general average of (positive= Improvement in QoL) 21% Routine Activity (4 possible responses for each question):Our category Routine Activity covers questions 6 to 19 of the QLQ-C30 questionnaire. Each of these questions has a range from 1 - 4, so the possible sum of the raw data ranges from 14 - 56, where 14 is the highest QoL (excellent QoL), and 56 is the lowest QoL (very poor QoL) In our Routine Activity category, we observed that 2 out of 19 patients replied that their QoL was impaired (10.5%) and 17 patients replied their QoL did improve (89.5%). The greatest improvement in QoL was 63.83%; and the greatest decrease in QoL was 25.00%; with a general average of (positive= Improvement in QoL) 30.98% Emotional Activity (4 possible responses for each question):Our category Emotional Activity covers questions 16 - 28 of the QLQ-C30 questionnaire. Each of these questions has a range from 1 - 4, so the possible sum of the raw data ranges from 13 - 52, where 13 is the highest QoL (excellent QoL), and 52 is the lowest QoL (very poor QoL). In our Emotional Activity category, we observed that 1 out of 19 patients replied that their QoL was impaired (5.2%) and 18 patients replied their QoL did improve (94.8%). The greatest improvement in QoL was 66.67%; and the greatest decrease in QoL was 6.25%; with a general average of (positive= Improvement in QoL) 33.22% General (Sum of Domestic, Routine & Emotional):A combination of the 3 categories Domestic, Routine and Emotional covers questions 1 to 28 of the QLQ-C30. The summed raw data ranges from 28 to 112, where 28 is the highest QoL (excellent QoL) and 112 is the lowest QoL (very poor QoL). We observed that 1 out of 19 patients in the study replied that their QoL was impaired (5.26%), none replied their QoL did not change at all, and 18 patients replied their QoL did improve (94.8%). The greatest improvement in Qol was 65.26%; and the greatest decrease in Qol was 2.08%; with a general average of (positive= Improvement in Qol) 31.49% Final QoL (Patient Score - 7 possible responses for each question):For questions 29 - 30 of the EORTC QLQ-C30 questionnaire, the possible score ranges from 2 to 14, where 14 is the highest QoL (excellent QoL) and 2 is the lowest QoL (very poor QoL) The greatest improvement in QoL was 500%; and the greatest decrease in QoL was 20.00%; with a general average of (positive= Improvement in QoL) 100.21% Table 2: Results. Sums of raw data for each patient in each category
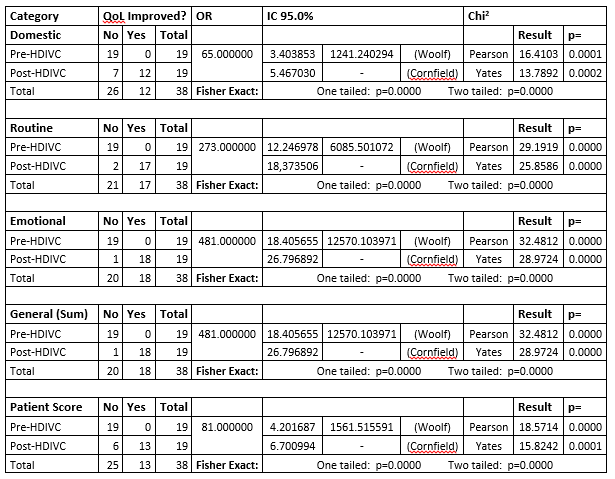
Pre = Pre HDIVC, Post = Post HDIVC, %? = percent change. A positive number for %? values in all categories means an improvement in QoL. STATISTICS ANALYSIS OF THE RAW DATA22 patients who fulfilled the requirements of inclusion criteria were recruited for the present study. 19 patients completed. The QLQ-C30 questionnaire was completed before and after doctors administered HDIVC to all 19 patients. A starting (base) line was used to compare the outcome of the QLQ-C30 questionnaire before and after the external doctor's administration of HDIVC (data not published). A Chi square Test was used for analysis, and for expected frequencies under 5, a Fisher exact test was used to compare qualitative variables. For our analysis, we selected as a Pre-HDIVC group: Patients on cancer allopathic therapies undergoing secondary effects and we selected as a Post-HDIVC group: the same Patients after HDIVC administration. For the Chi square and Fischer exact tests of association: HYPOTHESIS Megadose intravenous vitamin C significantly improves the quality of life in patients with any malignant cancer diagnosis who are receiving conventional medical treatments including chemotherapy, radiotherapy and/or hormone therapy; where these patients have secondary effects that may affect their quality of life; and are evaluated by the QLQ-C30 questionnaire. NULL HYPOTHESIS Megadose intravenous vitamin C does not affect the quality of life in patients with any malignant cancer diagnosis who are receiving conventional medical treatments including chemotherapy, radiotherapy and/or hormone therapy; and are evaluated by the QLQ-C30 questionnaire. A descriptive statistic was used to adjust the results of the QLQ-C30 questionnaire for each of the categories measuring changes in the quality of life (if there were any). The results were recorded as a percentage change, comparing the pre and post HDIVC raw scores for each patient in each category. The alfa error was 0.05 in all analyses. The statistics package EPIDAT 4.1 was used for all analyses. When zeros cause problems with calculation Epidat adds the standard error 0.5 to all cells. This happens because all our 2x2 tables have a "Pre-HDIVC" group containing 19 patients with secondary effects and 0 patients with absence of secondary effects. Table 3: Analysis of association between HDIVC administration and improvement in QoL
Association Chi squared results for all categories are above the critical value (for one degree of freedom) and all p values are < 0.05, therefore we reject the null hypothesis. Fisher Exact Test results for all categories have a p value < 0.05, therefore we reject the null hypothesis. In all categories administration of HDIVC showed a statistically significant association between HDIVC treatment and improvement in QoL scores. Domestic activity: Routine Activity: Emotional activity: Combined Domestic + Routine + Emotional Activities: Overall patient score: Our data suggests a strong association between HDIVC intervention and improved QoL in all categories. DISCUSSIONOurs is an observational study looking at the effect of HDIVC on the QoL of cancer outpatients receiving treatment in two integrative medicine centres in Bogota. HDIVC is commonly used in these centres with cancer patients who are also receiving standard therapies. The HDIVC doses used are more typical of the high doses being used in current clinical trials and aim to achieve high blood levels of Vit C. Many studies have reported improved QoL in cancer patients with HDIVC. Klimant et al. (12) in 2018 have reviewed the use of HDIVC in cancer treatments and give recommendations for an effective but conservative use of HDIVC in combination with chemotherapy. Studies investigating the effect of HDIVC, including QoL studies, have used a large range of doses from as little as 7.5 g up to 150 g of IVC with a variety of dosing schedules employed. However very few studies have been formal QoL studies measuring QoL using a variety of validated questionnaires. Some other previous QoL studies have examined lower dose IVC; doses of 7.5 grams weekly (18) and 10 grams (2 doses 3 days apart) (21), doses that are much lower than the typical dose being used in current clinical trials and the Bogota medical centres. Three other QoL studies have examined a higher dosage schedule. Ou et al. in 2017 (19) used 1.0, 1.2 or 1.5 grams/kg, Takahashi et al (20) in 2011 observed the Riordan protocol, approx. 140 grams/week, and Bazzan et al. (22) in 2018 observed a wide range of doses and schedules in a hospital, ranging from 50 - 150 grams per dose). Three previous studies (Ou et al. (19) high dose, Takahashi et al (20) high dose and Yeom et al (21) lower dose) have used the QLQ-30 for measuring pre and post HDIVC in cancer patients. Our results compare favourably with results from previous QLQ-30 studies. Table 4: EORTC QLQ-30, comparison of global health status
Our results also compare favourably with studies that have used different methods for comparing QoL (Vollbracht et al 2011 (18), Bazzan et al (22) 2018) Ours is not a dose ranging study. We have not asked the medical centres to modify their treatments, QoL is not their only reason for treatment. We have not collected data on the effects of different doses or HDIVC protocols on QoL. The Riordan protocol used in the USA (24) is titrated for each patient to achieve a 350 mg% blood level of Vit C at around day 3 of the protocol. Some patients require considerably more IVC than average to reach these levels, some require considerably less. So, the total amount of Vit C administered over the period of the protocol can vary quite a bit from patient to patient. In clinical experience in Colombia, the amount of HDIVC required to achieve high blood levels is on average lower than that in the USA. Also, the Bogota medical centres routinely test for G6PD deficiency and adequate kidney function in all patients prior to IVC treatment as outlined in the Riordan protocol (24). G6PD deficiency is uncommon in our Colombian population and appears to be more common in the USA and some other countries. We are aware then of some differences in our population compared to G6PD levels in the USA. LIMITATIONS OF THE STUDYThis is an observational study. Associations have been measured between high dose Vitamin C administration and changes in quality of life. We have sought to gain preliminary data for the effectiveness of Vitamin C administration in our population, with a view to adequately sizing a future clinical trial into Vitamin C and QoL in cancer patients. This study has not answered the open questions remaining about the use of high dose Vitamin C in QoL, we have not observed or measured: the effect of dose ranging on different cohorts, the duration of effectiveness of a dose, the use of a placebo control. CONCLUSIONWe have had encouraging results from our observational study. In both the EORTC QLQ-30 scores and a percent change descriptive statistic we observed that HDIVC was associated with a significant improvement in reported QoL. For the QLQ-30 scale scores significant improvements in QoL (p < 0.01) were observed for; Global health status/QoL, Physical functioning, Role functioning, Emotional functioning, Social functioning, Fatigue, Pain, Insomnia and significant improvements in QoL (p < 0.05) were observed for: Nausea and vomiting, Dyspnoea, Appetite loss. Out of the 19 patients evaluated in the categories Domestic Activity, Routine Activity and Emotional Activity, only one did not report improvement in their quality of life and 18 patients reported improvement (94.73%). All statistical tests in these categories showed a strong association between HDIVC treatment and improvement of QoL in our study population. Overall the administration of HDIVC had a significant positive effect on patients' quality of life. Further trials into HDIVC on QoL in cancer cohorts are warranted. List of AbbreviationsEORTC: European Organisation for Research and Treatment of Cancer
Consent for publicationThis paper is presented on the Orthomolecular Medicine News Service with the kind permission of the Australasian College of Nutritional and Environmental Medicine (ACNEM) https://www.acnem.org/ , who first published it in ACNEM Journal 2109, 38:4, p 28-37. All data has been de-identified. Individual patients are referred to by an identification number or patient code. Patients have signed a consent form for publication. No patient identifiable data is included in the publication or associated tables. Competing InterestsHG has received research grants from Biological Therapies comprising of IVC vials supplied free of charge for this study. HG owns stock in Drug company Grupo Gales SAS which imports vitamin C from Biological Therapies to Colombia. CC declares that he has no competing interest. GG declares that she has no competing interest. FundingBiological Therapies, AUSTRALIA, A Division of Orthomolecular Medisearch Laboratories Pty Ltd. A.C.N. 006 897 856 Suite 5, 20-30 Malcolm Road (PO Box 702) Braeside VIC 3195 Australia Biological Therapies provided free of charge the Vitamin C used in this study, registered in Colombia INVIMA 2016M-0012358-R1 Author's contributionsHG, CC and GG designed the study. CC prepared the data tables for the study and conducted the analyses once data was complete at patient no. 22. CC prepared the interpretations of the data in Spanish. HG prepared the original manuscript and translated all materials from Spanish into English. This study has not been published in Spanish. AcknowledgementsLOCAL ASSISTANTS Mr. Jhon Cantor, Statistical Analysis Assistant, Grupo Gales
JC, AL, MPP and SV provided logistics and support for the study. EXTERNAL DOCTORS Dr. Helber Prieto Epidemiologist, Specialist in Occupational Health/Medicine, SCMPO (The Colombian Society of Preventive Medicine).
HP, SR and RL were observed in clinical practice. HP provided support for statistics analysis Authors:Dr. Hugo Galindo Salom, Epidemiologist Specialist, Master's in education SCMPO (Sociedad Colombiana de Medicina Preventiva y Ortomolecular - The Colombian Society of Preventive Medicine). https://www.sociedadcolombianamedicinapreventiva.com/ Via Cajica Km 1.5 Avenida Los Zipas Tres Esquinas Sector Bomberos Edificio Conex, Consultorio 610 Tel: (571) 795 4215 Chía Colombia hgalindo@grupogales.com Dr. Carlos Carrillo, Epidemiologist Specialist, SCMPO (Sociedad Colombiana de Medicina Preventiva y Ortomolecular - The Colombian Society of Preventive Medicine). https://www.sociedadcolombianamedicinapreventiva.com/ Via Cajica Km 1.5 Avenida Los Zipas Tres Esquinas Sector Bomberos Edificio Conex, Consultorio 610 Tel: (571) 795 4215 Chía Colombia bravoscarlos04@gmail.com Prof. Gladys Galvis, Alternative Therapies Co-ordinator, Rosario University, Colombia. glagalvis@gmail.com References1. Weber P, Bendich A, Schalch W. (1996) Vitamin C and human health--a review of recent data relevant to human requirements. Int J Vitam Nutr Res Int Z Vitam- Ernahrungsforschung J Int Vitaminol Nutr. 66:19-30. https://pubmed.ncbi.nlm.nih.gov/8698541 2. Peterkofsky B. (1991) Ascorbate requirement for hydroxylation and secretion of procollagen: relationship to inhibition of collagen synthesis in scurvy. Am J Clin Nutr. 54(6 Suppl):1135S-1140S. https://pubmed.ncbi.nlm.nih.gov/1720597 3. McRae MP. (2008) Vitamin C supplementation lowers serum low-density lipoprotein cholesterol and triglycerides: a meta-analysis of 13 randomized controlled trials. J Chiropr Med. 7:48-58. https://pubmed.ncbi.nlm.nih.gov/19674720 4. Padayatty SJ, Levine M. (2016) Vitamin C physiology: the known and the unknown and Goldilocks. Oral Dis. 22:463-493. https://onlinelibrary.wiley.com/doi/epdf/10.1111/odi.12446 5. Camarena V, Wang G. (2016) The epigenetic role of vitamin C in health and disease. Cell Mol Life Sci. 73:1645-58. https://pubmed.ncbi.nlm.nih.gov/26846695 6. Marik PE. (2018) Hydrocortisone, Ascorbic Acid and Thiamine (HAT Therapy) for the Treatment of Sepsis. Focus on Ascorbic Acid. Nutrients. 10(11). https://www.mdpi.com/2072-6643/10/11/1762 7. Kahn SA, Beers RJ, Lentz CW. (2011) Resuscitation After Severe Burn Injury Using High-Dose Ascorbic Acid: A Retrospective Review: J Burn Care Res. 32:110117. https://pubmed.ncbi.nlm.nih.gov/21131846 8. Ichim TE, Minev B, Braciak T, Luna B, Hunninghake R, Mikirova NA, et al. (2011) Intravenous ascorbic acid to prevent and treat cancer-associated sepsis? J Transl Med. 9:25. https://translational-medicine.biomedcentral.com/track/pdf/10.1186/1479-5876-9-25 9. Lane DJR, Richardson DR. (2014) The active role of vitamin C in mammalian iron metabolism: Much more than just enhanced iron absorption! Free Radic Biol Med. 75:69-83. https://pubmed.ncbi.nlm.nih.gov/25048971 10. Casciari JJ, Riordan NH, Schmidt TL, Meng XL, Jackson JA, Riordan HD. (2001) Cytotoxicity of ascorbate, lipoic acid, and other antioxidants in hollow fibre in vitro tumours. Br J Cancer. 84:1544-1550. https://pubmed.ncbi.nlm.nih.gov/11384106 11. Carr AC, Vissers MCM, Cook JS. (2014) The Effect of Intravenous Vitamin C on Cancer- and Chemotherapy-Related Fatigue and Quality of Life. Front Oncol 4:283 . https://www.frontiersin.org/articles/10.3389/fonc.2014.00283/full 12. Klimant E, Wright H, Rubin D, Seely D, Markman M. (2018) Intravenous vitamin C in the supportive care of cancer patients: a review and rational approach. Curr Oncol. 25:139-148. https://www.current-oncology.com/index.php/oncology/article/view/3790/2697 13. Jacob RA, Sotoudeh G. (2002) Vitamin C function and status in chronic disease. Nutr Clin Care 5:66-74. https://pubmed.ncbi.nlm.nih.gov/12134712 14. Cameron E, Pauling L. (1976) Supplemental ascorbate in the supportive treatment of cancer: Prolongation of survival times in terminal human cancer. Proc Natl Acad Sci. 73:3685-3689. https://pubmed.ncbi.nlm.nih.gov/1068480 15. Parrow NL, Leshin JA, Levine M. (2013) Parenteral Ascorbate As a Cancer Therapeutic: A Reassessment Based on Pharmacokinetics. Antioxid Redox Signal. 19:2141-2156. https://pubmed.ncbi.nlm.nih.gov/23621620 16. Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, et al. (1993) The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 85:365-376. https://pubmed.ncbi.nlm.nih.gov/8433390 17. Cruz Bermudez HF, Moreno Collazos JE, Angarita Fonseca A (2013). Medición de la calidad de vida por el cuestionario QLQ-C30 en sujetos con diversos tipos de cáncer de la ciudad de Bucaramanga- Colombia. Enferm Glob 12 (2): #30 - Abril . http://revistas.um.es/eglobal/article/view/eglobal.12.2.160351 18. Vollbracht C, Schneider B, Leendert V, Weiss G, Auerbach L, Beuth J. (2011) Intravenous Vitamin C Administration Improves Quality of Life in Breast Cancer Patients during Chemo-/Radiotherapy and Aftercare: Results of a Retrospective, Multicentre, Epidemiological Cohort Study in Germany. In Vivo. 25:983-990. http://iv.iiarjournals.org/content/25/6/983.long 19. Ou J, Zhu X, Lu Y, Zhao C, Zhang H, Wang X, et al. (2017) The safety and pharmacokinetics of high dose intravenous ascorbic acid synergy with modulated electrohyperthermia in Chinese patients with stage III-IV non-small cell lung cancer. Eur J Pharm Sci. 109:412-418. https://pubmed.ncbi.nlm.nih.gov/28847527 20. Takahashi H, Mizuno H, Yanagisawa A. (2012) High-dose intravenous vitamin C improves quality of life in cancer patients. Pers Med Universe. 1:49-53. https://www.sciencedirect.com/science/article/abs/pii/S2186495012000132 21. Yeom CH, Jung GC, Song KJ. (2007) Changes of Terminal Cancer Patients? Health-related Quality of Life after High Dose Vitamin C Administration. J Korean Med Sci. 22:7-11. https://pubmed.ncbi.nlm.nih.gov/17297243 22. Bazzan AJ, Zabrecky G, Wintering N, Newberg AB, Monti DA. (2018) Retrospective Evaluation of Clinical Experience With Intravenous Ascorbic Acid in Patients With Cancer. Integr Cancer Ther. 17:912-920. https://journals.sagepub.com/doi/10.1177/1534735418775809 23. Riordan HD, Hunninghake RB, Riordan NH, Jackson JJ, Meng X, Taylor P, et al. (2003) Intravenous ascorbic acid: protocol for its application and use. P R Health Sci J. 22:287-290. https://riordanclinic.org/wp-content/uploads/2014/12/89022715.pdf 24. Riordan Clinic (2013) The Riordan IVC Protocol for Adjunctive Cancer Care. Intravenous Ascorbate as a Chemotherapeutic and Biological Response Modifying Agent. Riordan Clinic. https://riordanclinic.org/research-study/vitamin-c-research-ivc-protocol Nutritional Medicine is Orthomolecular MedicineOrthomolecular medicine uses safe, effective nutritional therapy to fight illness. For more information: http://www.orthomolecular.org Find a DoctorTo locate an orthomolecular physician near you: http://orthomolecular.org/resources/omns/v06n09.shtml The peer-reviewed Orthomolecular Medicine News Service is a non-profit and non-commercial informational resource. Editorial Review Board:
Ilyès Baghli, M.D. (Algeria)
Andrew W. Saul, Ph.D. (USA), Editor-In-Chief
Comments and media contact: drsaul@doctoryourself.com OMNS welcomes but is unable to respond to individual reader emails. Reader comments become the property of OMNS and may or may not be used for publication. Click here to see a web copy of this news release: https://orthomolecular.activehosted.com/p_v.php?l=1&c=171&m=175&s=ba4ddd4a14f7aaee0907647bc30f1d93 |
|||||||||||||||||||||||
|
This news release was sent to chris.pedersen@nowfoods.com. If you no longer wish to receive news releases, please reply to this message with "Unsubscribe" in the subject line or simply click on the following link: unsubscribe . To update your profile settings click here . This article may be reprinted free of charge provided 1) that there is clear attribution to the Orthomolecular Medicine News Service, and 2) that both the OMNS free subscription link http://orthomolecular.org/subscribe.html and also the OMNS archive link http://orthomolecular.org/resources/omns/index.shtml are included.
|
|||||||||||||||||||||||